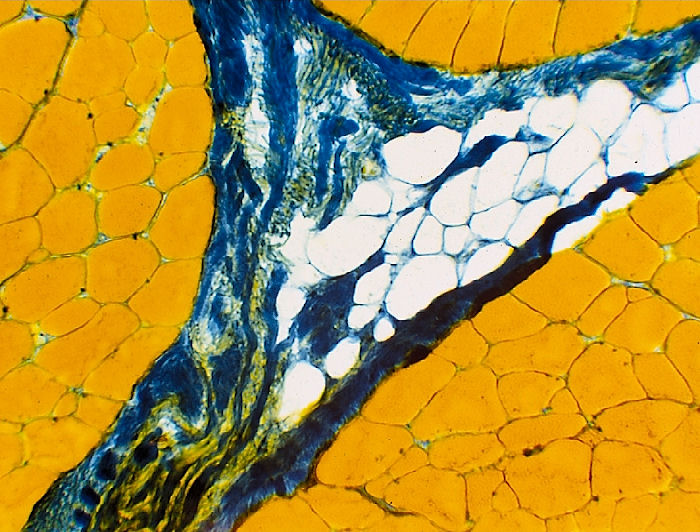

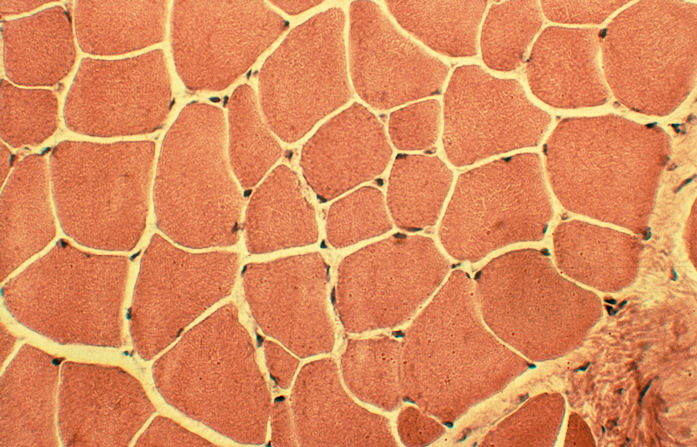
Hematoxylin - Eosin Stain (H&E)
H&E is a
good general stain, staining nucleic acids in the nucleus blue and the
cytoplasm pink.
1. Cut tissue sample on a microtome 10 to 12
µm
thick and place onto microscope slides (microscope slides should be
pre-rinsed/cleaned in an ethanol solution). Leave slides at room temperature
for at least 5 min.
2. Fix sections in 4% formaldehyde-Ca2+ (see notes) for 3-5 min.
3. Rinse for 5 min in dH2O.
4. Stain sections with Hematoxylin (Haemalaun acc. Meyer) for 5 min.
5. Rinse continuously under tap water for 20 min to rinse off remaining
Hematoxylin stain.
6. Rinse for 5 min in d2H2O.
7. Stain sections with a 0.1 % solution of Eosin (Potassium salt) in dH2O
for 10 min.
8. Rinse for 3 min in dH2O.
9. Wash with:
70% Ethanol for 1 min
96% Ethanol for 1 min
absolute Ethanol for 3 min
10. Fix with Xylol for between 5 and 60 min (dispose of Xylol in a proper
receptacle after use).
11. Mount a cover slip using a non-aqueous cover slip medium (ie. Histofluid).
Notes:
- To prepare the 4% Formaldehyde solution you add 1 g CaCl2 (water-free - VERY important) for every 100 mL of 4% formaldehyde. A 4% formaldehyde solution can be made from stock 37% Formalin by dilution. To the CaCl2/4% formaldehyde solution you add CaCO3. You need to add enough so that it does not dissolve and it is visible at the bottom of the jar. Basically the solution has to be supersaturated. In our solution you can see about 2 mm of CaCO3 on the bottom of the jar in a slurry. Just make sure to filter the solution before using it. The CaCO3 will go into solution so make sure that you add enough. It should settle to the bottom after awhile.
- The hematoxylin solution is purchased
pre-mixed. We use Mayers Hematoxylin.
The 0.1% Eosin solution is made with water. Simply mix 0.1 g of Eosin in 100
mL of water.
-
Unless otherwise noted solutions are to be kept at 4 °C, are good for up to
one month, and can
be disposed of down the sink.